|
6/10/2023 0 Comments Nuclear envelope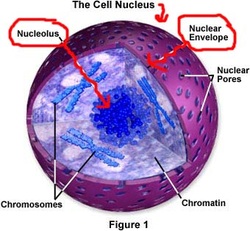
(2004) concluded that CPG2 is a component of a specialized postsynaptic endocytic mechanism devoted to internalization of synaptic proteins, including glutamate receptors. Manipulating Cpg2 levels also affected dendritic spine size. (2004) found that RNA interference-mediated knockdown of Cpg2 in cultured rat hippocampal neurons increased the number of postsynaptic clathrin-coated vesicles, some of which trafficked NMDA-type glutamate receptors (see GRIN1 138249), disrupted constitutive internalization of glutamate receptors, and inhibited activity-induced internalization of synaptic AMPA-type glutamate receptors (see GRIA1 138247). When the transmembrane domain was deleted, the remaining C-terminal domain directed nuclear targeting.Ĭottrell et al. (2001) determined that SYNE1 requires its C-terminal transmembrane domain to localize to the nuclear envelope. Nesprin transcription appeared to be highly adaptable, with a feedback loop regulating variant expression, as perturbing expression of 1 transcript influenced expression of other downstream transcripts. Expression analysis in human fibroblasts and U2OS cells showed that subcellular localization of these variants depended on cell type. Both nesprin-1 and nesprin-2 underwent alternative splicing and expressed multiple tissue-specific variants generated by alternate initiation and termination. (2012) identified multiple nesprin-1 and nesprin-2 (SYNE2 608442) variants. Immunohistochemical analysis localized Cpg2 to the postsynaptic side of excitatory synapses on rat glutamatergic neurons.īy database and RACE analyses, followed by PCR and DNA sequencing, Rajgor et al. RT-PCR detected the 2 Cpg2 transcripts only in rat brain. Compared with Cpg2, Cpg2b has an additional exon at its 5-prime end. The 3-prime UTR of Cpg2 contains an unspliced intron between exons 33 and 34 of the Syne1 gene, and exon 34 is followed by a noncanonical polyadenylation hexamer that is conserved in rats and humans. (2004) identified 2 brain-specific splice variants of rat Syne1, which they called Cpg2 and Cpg2b, that encode proteins of 941 and 965 amino acids, respectively. (2002) generated antibody to the newly identified N-terminal portion of SYNE1 and showed that SYNE1 localized to the sarcomeres of cardiac and skeletal muscle.Ĭottrell et al. 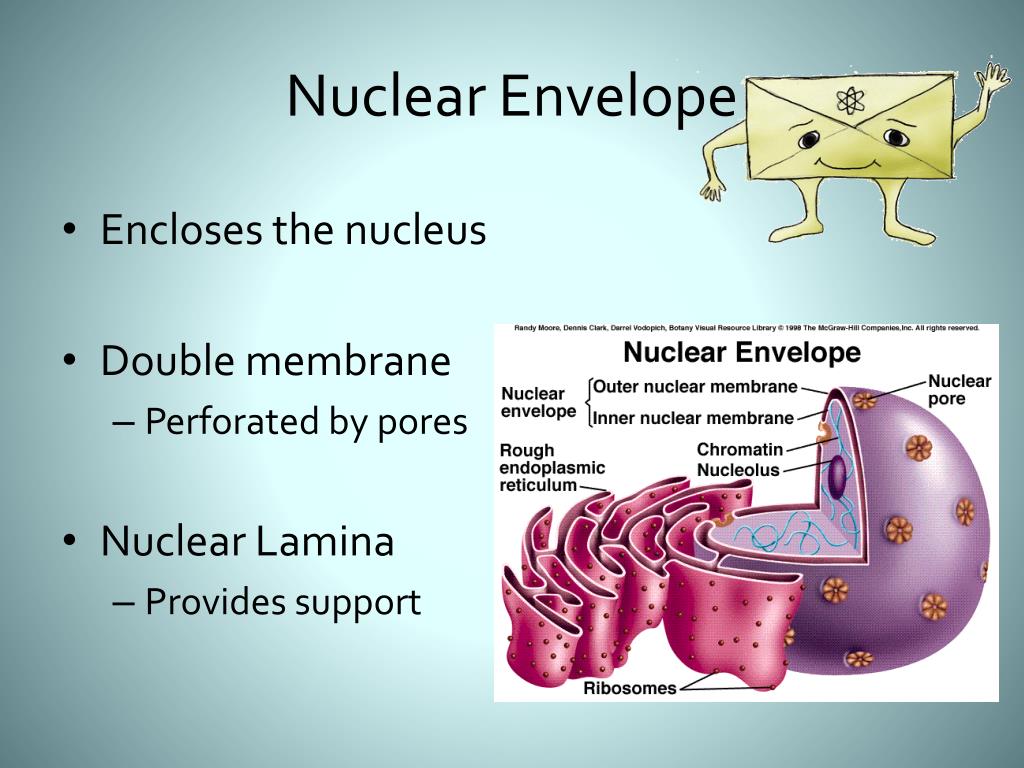
They also identified SYNE1 homologs in Drosophila and C. (2002) extended the sequence of SYNE1 and showed that it encodes an 8,797-amino acid protein with an actin-binding region containing 2 tandem calponin (see 600806) homology domains at its N terminus. Immunogold and immunofluorescence analyses localized nesprin-1 primarily to the nuclear envelope, with occasional nuclear staining showing colocalization with heterochromatin and the nucleolus.īy genomic sequence analysis, RT-PCR, and RACE, Zhang et al. Northern blot analysis detected ubiquitous expression of 3.8- and 10.7-kb transcripts, with highest levels in spleen, peripheral blood leukocytes, and heart. Nesprin-1-beta also contains a protein-DNA binding motif. Both nesprin-1 proteins contain multiple spectrin repeats, a bipartite nuclear localization signal, a transmembrane domain within the C-terminal Klarsicht homology (KASH) region, and several N-glycosylation and phosphorylation sites. They also identified a rare splice variant of nesprin-1-alpha, nesprin-1-alpha-2, that contains an additional 31 amino acids at its N terminus. Nesprin-1-alpha contains 982 amino acids and nesprin-1-beta contains 3,221 amino acids. (2001) isolated human cDNAs encoding 2 SYNE1 proteins, which they called nesprin-1-alpha and nesprin-1-beta. By searching sequence databases using rat Syne1 as probe, followed by PCR and RACE of human spleen and heart cDNA libraries, Zhang et al. (2001) obtained a rat Syne1 cDNA that was upregulated in differentiated aortic vascular smooth muscle cells. In skeletal muscle, high levels of expression were detected in nuclei associated with synaptic sites. Immunofluorescence on adult mouse tissues localized Syne1 to nuclei in skeletal, smooth, and cardiac muscle cells. RT-PCR analysis showed moderate to high expression of both transcripts in most human tissues examined. Northern blot analysis of mouse and human tissues detected a 4.7-kb SYNE1A transcript in skeletal and cardiac muscle and a 10-kb SYNE1B transcript in multiple tissues, including heart, muscle, kidney, liver, and brain. (2000) identified KIAA0796 ( Nagase et al., 1998) as a partial human ortholog of Syne1. Syne1 contains multiple spectrin repeats and a 60-amino acid C-terminal region homologous to the Drosophila protein Klarsicht. By screening additional mouse cDNA libraries, they identified cDNAs encoding at least 3 Syne1 variants. (2000) isolated a cDNA encoding Syne1 by yeast 2-hybrid analysis of an embryonic mouse cDNA library using mouse Musk ( 601296) as bait.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
